[ad_1]
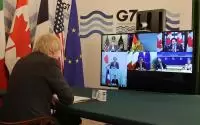
COVAX, the push to equitably distribute COVID-19 vaccine throughout the world, got a major boost today with a fresh infusion of pledges, as G7 leaders gathered virtually at a security conference.
In other international developments, Johnson & Johnson today submitted its COVID-19 vaccine for World Health Organization (WHO) assessment, and two researchers published more promising findings for already listed vaccines, AstraZeneca-Oxford and Pfizer-BioNTech.
Table of Contents
G7 support adds more momentum to COVAX
In advance of the G7 meeting, who convened the group, encouraged the participants from some of the biggest economies in the world to sign onto a new WHO declaration on vaccine equity.
At today’s meeting, leaders from the Canada, France, Germany, Italy, Japan, the European Union, the United Kingdom, and the United States issued a joint statement intensifying cooperation on the pandemic response, which included pledging more than $4 billion more to COVAX, a key step toward reducing the global threat from the virus by easing access to vaccine to all countries, especially countries without the resources to develop or buy their own supplies. The new funding pushes the group’s support to $7.5 billion.
US President Joe Biden, in his first G7 appearance, pledged an initial $2 billion for Gavi, the Vaccine Alliance, which finances vaccine, including through COVAX, for developing countries. The money was appropriated by Congress in December. The Biden Administration also said it would release another $2 billion through 2021 and 2022, with the first $500 million made available when current donors fulfil their pledges and the first doses are delivered to countries.
In another major announcement, the European Union today announced that it was doubling its contribution to COVAX, from €500 million ($606 million) to €1 billion ($1.2 billion). Ursala von der Leyden, president of the European Commission, said today that the EU is also providing €100 million ($121 million) to support the rollout of vaccine in Africa in a partnership with Africa Centers for Disease Control and Prevention.
WHO welcomes vaccine equity support
The WHO, which has led the COVAX effort along with several partners including the Coalition for Epidemic Preparedness Innovations (CEPI), today praised the major new boost for COVAX. Its Director-General Tedros Adhanom Ghebreyesus, PhD, said in a statement, “There is a growing movement behind vaccine equity and I welcome that world leaders are stepping up to the challenge by making new commitments to effectively end this pandemic by sharing doses and increasing funds to COVAX.”
Also, the WHO said nearly 7,000 people and groups have signed its new vaccine equity declaration, which pushes governments and vaccine makers to speed the regulatory process, beef-up manufacturing, and share technology.
The first vaccine doses through COVAX for lower- to middle-income countries will start shipping at the end of the month, and the program’s goal is to distribute 350 million doses of AstraZeneca-Oxford vaccine in the first half of the year.
Other global headlines
- Johnson & Johnson announced today that it has submitted its materials to the WHO for its advisory committee to assess for emergency listing, a key step that paves to way for distribution to developing countries. The company’s vaccine requires only one dose.
- India is reporting an uptick in cases, which are at a 3-week high, with most reported in Kerala state and Maharashtra state, which includes Mumbai, according to Reuters. Officials said the reopening of schools in Kerala state and the resumption of suburban train service around Mumbai may be fueling the recent rise.
- The AstraZeneca-Oxford vaccine has higher efficacy when given with a 3-month interval between the two doses than with the original 6-week interval, Oxford-based researchers reported today in The Lancet. The team had earlier posted its findings in a preprint study. Based on phase 3 data analysis, they found an overall efficacy of 81% with the longer interval, compared to 55% with the shorter interval.
- Preprint study findings for the Pfizer-BioNTech vaccine from Israel’s health ministry suggest it may play a role in cutting transmission, according to national database findings from the country’s vaccine rollout, first reported by Israel’s Ynet news and obtained by Reuters. The results suggest the vaccine was linked to an 89.4% drop in asymptomatic cases and a 93.7% decline in symptomatic infections.
- The global total today rose to 110,562,484 cases, and 2,448,188 people have died from their infections, according to the Johns Hopkins online dashboard.
[ad_2]
Source link