[ad_1]
In the latest international COVID-19 developments, South Africa health officials announced they will share the country’s AstraZeneca-Oxford vaccine with other African nations, and in Europe, Johnson & Johnson submitted its request for an emergency use authorization for its single-dose COVID-19 vaccine.
Table of Contents
South Africa switch includes study of J&J vaccine
South Africa recently announced a pause on the rollout of AstraZeneca-Oxford vaccine, following early study findings that it appears have little impact on mild-to-moderate disease from the B1351 variant that is dominant in the country.
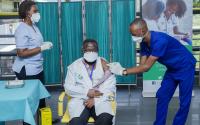
It followed that development with an announcement that it would temporarily switch to using the unapproved Johnson & Johnson vaccine. As part of a research study, the country will begin vaccinating health workers with the Johnson & Johnson vaccine, according to Reuters, with 80,000 doses expected initially, and 500,000 health workers targeted for immunization.
Anban Pillay, with South Africa’s health department, told Reuters that the country plans to share—with the help of the African Union—1 million doses of AstraZeneca-Oxford vaccine that was produced at the Serum Institute of India.
The WHO yesterday announced that it has officially listed the AstraZeneca-Oxford vaccine made in South Korea and India for emergency use, clearing the way for lower and middle income countries to receive supply through COVAX.
In a statement today, Gavi, the Vaccine Alliance, and other groups involved in COVAX welcomed the news and said they can now finalize their allocations for countries, which will be announced on Feb 22. Participating countries need to take their final steps, which include giving regulatory approval, signing indemnity agreements, and submitting their vaccine deployment plans. Those that have already completed those steps are likely to begin receiving doses later this month, with the bulk of deliveries to come in March.
COVAX partners have said they expect to distribute 350 million doses in the first half of the year.
EMA weighs Johnson & Johnson vaccine
In other vaccine developments today, the European Medicines Agency (EMA) today announced that it has received an application for emergency use authorization from Janssen-Cilag International for the Johnson & Johnson COVID-19 vaccine. The product is the fourth COVID-19 vaccine to be considered by the EMA, following the authorizations of the ones from Pfizer-BioNTech, Moderna, and AstraZeneca-Oxford.
The EMA said it will assess the vaccine on an accelerated timetable and that it has already reviewed some data during the rolling review process. It added that it is now assessing more data on efficacy, safety, and quality.
Also, Novavax today announced that it has expanded an agreement with SK Biosciences in South Korea to make and market 40 million doses of the vaccine for the South Korean market.
In other global headlines:
- Due to steady declines in cases, some governments announced plans to ease some restrictions, including Israel, which is making steady progress vaccinating its adult population, and Hong Kong, where cases have dropped to the lowest levels since November.
- In Europe, a Dutch court struck down the Netherlands’ COVID-19 curfew, ruling that the government lacks the authority, according to Reuters, which said the measure will remain in place during the appeal process. Also, German officials in the city of Hamm are doing mass testing after a B117 variant case was detected at an apartment building complex, as European Union officials announced a new regional collaboration to study called the “HERA incubator” to study mutations in SARS-CoV-2.
- South Korea’s intelligence agency said today that computer hackers in North Korea tried to obtain information on the COVID-19 vaccine from Pfizer-BioNTech, according to Reuters.
- The global total today is at 109,377,180 cases, and 2,414,570 people have died from their infections, according to the Johns Hopkins online dashboard.
[ad_2]
Source link